 It is the 8th most abundant element in the Earth's crust, constituting 2% by mass. The highest oxidation state of an element is determined using the periodic table by the group in which it is located. This enables the metals to easily lose electrons, which increases their stability and allows them to form compounds via ionic bonds. The charge of the monatomic ion is nothing but the charge of the ion. Antoine Bussy was the first to produce it in consistent form in 1831. All the elements in Group 2 have two electrons in their valence shells, giving them an oxidation state of +2. The oxidation number of any free element is always 0. Magnesium was first discovered in 1808 by Sir Humphry Davy in England by the electrolysis of magnesia and mercury oxide. The image is optimized to function as a widescreen wallpaper with a resolution of 3840 x 2160. The different colors correspond to the different element groups with a key along the bottom. Values in italics represent theoretical or unconfirmed oxidation numbers. Each element square contains all 118 of elements with the element number, symbol, name, atomic mass, and most common oxidation number. Bold numbers represent the more common oxidation states. Atomic Number = 12 Mass = 24.31 g mol -1 Electrion Configuration = 3s 2 Density = 1.738 g cm -3 This periodic table contains the oxidation numbers of the elements.Its four oxidation states represent local minima, and moving between them requires an activation energy, making each of the states relatively stable, but possible to dislodge, i.e. One reason for this is that while iron atoms have a " the lowest energy state", it's difficult to get at without first coming across several " a (locally) lowest energy state". : The oxidation number of Zn, Al, H2, O2, and Cl2 is zero : The oxidation number of Na+ is +1, Mg2+ is +2, Al3+ is +3, Cl-1 is -1, and O2- is -2. At 26, iron is a lightweight, but already there you have 4 (common!) oxidation states, the two most common ones being iron(II) and iron(III). In this case we have to consider the breaking of all covalent bonds and. The reason behind this is that the element has neither lost any electrons, nor gained any, and is therefore neutral. The oxidation state of the element will be the hypothetical charge if it were an ion. Period 2 is the first period in the periodic table from which periodic trends can be drawn. The oxidation number of all uncombined elements is 0. Registered charity number: 207890 Facebook Twitter. Periodic trends Calculated atomic radii of period 2 elements in picometers. The Royal Society of Chemistrys interactive periodic table features history, alchemy, podcasts, videos, and data trends across the periodic table. The oxidation number of H is +1, but it is -1 in when combined with less electronegative elements. 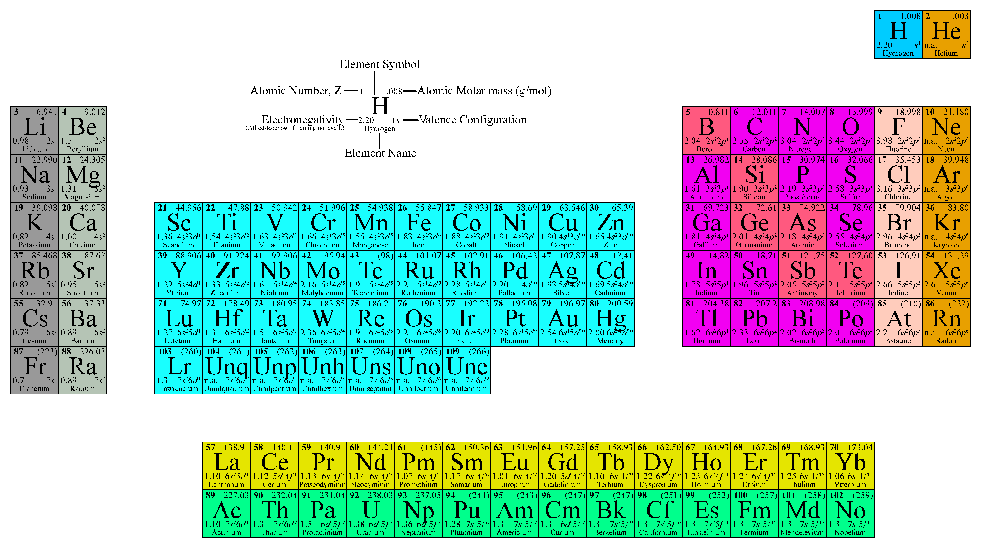
The oxidation number of a monatomic ion equals the charge of the ion. The oxidation number of a free element is always 0. 
The truth, as always, is a little more complicated:Ĭompared to the (somewhat) well-behaved non-metals in the upper right corner of the periodic table, most metals have a lot for electrons. Interactive periodic table with element scarcity (SRI), discovery dates, melting and boiling points, group, block and period information. Answer (1 of 2): The general rules for oxidation numbers are: 1. In this particular case, you've probably been taught about the octet rule, a rule which makes it seem as if any given element should have exactly two oxidation states 'n' and '8-n', where n is a simple function of it's location in the periodic table. Because a lot of what you have been taught in school is a lie. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
